Advertisements
Advertisements
प्रश्न
Give an example for a complex of the type [Ma2b2c2] where a, b, c are monodentate ligands and give the possible isomers.
उत्तर
The octahedral complexes of [Ma2b2c2] type can exist in five geometrical isomers.
The five geometrical isomers for the complex ion [PtCl2(NH3)2(py)2]2+are shown below.
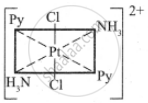
(I)
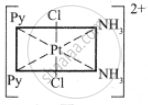
(II)
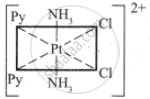
(III)
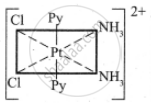
(IV)
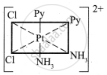
(V)
APPEARS IN
संबंधित प्रश्न
A complex has a molecular formula \[\ce{MSO4Cl.6H2O}\] The aqueous solution of it gives white precipitate with Barium chloride solution and no precipitate is obtained when it is treated with silver nitrate solution. If the secondary valence of the metal is six, which one of the following correctly represents the complex?
Crystal field stabilization energy for high spin d5 octahedral complex is ____________.
Arrange the following in order of increasing molar conductivity.
i) \[\ce{Mg[Cr(NH3) (Cl)5]}\]
ii) \[\ce{[Cr(NH3)5 Cl]3 [CoF6]2}\]
iii) \[\ce{[Cr(NH3)3 Cl3]}\]
In an octahedral crystal field, draw the figure to show the splitting of d orbitals.
Why tetrahedral complexes do not exhibit geometrical isomerism.
What is crystal field splitting energy?
What is crystal field stabilization energy (CFSE)?
A solution of \[\ce{[Ni(H2O)6]^2+}\] is green, whereas a solution of \[\ce{[Ni(CN)4]^2-}\] is colorless – Explain.
What are the limitations of VB theory?
Write the oxidation state, coordination number, nature of ligand, magnetic property and electronic configuration in octahedral crystal field for the complex \[\ce{K4[Mn(CN)6]}\].
