Advertisements
Advertisements
प्रश्न
Give an expression for work done in an isothermal process.
उत्तर
The work done by the gas, W = `int_("V"_"i")^("V"_"f") "P"."dV"`
Writing pressure in terms of volume and temperature,
P = `(μ"RT")/"V"`
Substituting this value we get
W = `int_("V"_"i")^("V"_"f") (μ"RT")/"V" "dV" = μ"RT" int_("V"_"i")^("V"_"f") "dV"/"V"`
By performing the integration in equation, we get
W = `μ"RT" ln ("V"_"f"/"V"_"i")`
APPEARS IN
संबंधित प्रश्न
Give an example of some familiar process in which heat is added to an object, without changing its temperature.
Draw a p-V diagram showing positive work at constant pressure.
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
Write a note on free expansion.
Explain the thermodynamics of the isobaric process.
The V-T diagram of an ideal gas which goes through a reversible cycle A→B→C→D is shown below. (Processes D→A and B→C are adiabatic)
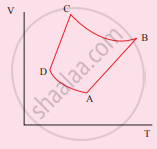
The corresponding PV diagram for the process is (all figures are schematic)
Draw the PV diagram for the isochoric process.
Draw the TP diagram (P-x axis, T-y axis), VT(T-x axis, V-y axis) diagram for
- Isochoric process
- Isothermal process
- Isobaric process
For a given ideal gas 6 × 105 J heat energy is supplied and the volume of gas is increased from 4 m3 to 6 m3 at atmospheric pressure. Calculate
- the work done by the gas
- change in internal energy of the gas
- graph this process in PV and TV diagram
Give any two types of a thermodynamic process.
