Advertisements
Advertisements
प्रश्न
Give the equation of state for an isothermal process.
उत्तर
It is a process in which the temperature remains constant but the pressure and volume of a thermodynamic system will change. The ideal gas equation is PV = µRT
APPEARS IN
संबंधित प्रश्न
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
Explain graphically (i) positive work with varying pressure, (ii) negative work with varying pressure, and (iii) positive work at constant pressure.
In an isochoric process, we have ____________.
Give an expression for work done in an isothermal process.
Two identical samples of a gas are allowed to expand (i) isothermally (ii) adiabatically. Work done is ____________.
Which of the following processes is reversible?
Give any two types of a thermodynamic process.
In a certain thermodynamical process, the pressure of a gas depends on its volume as kV3. The work done when the temperature changes from 100°C to 300°C will be ______ nR, where n denotes number of moles of a gas.
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.
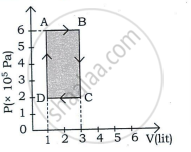
In a cyclic process, if ΔU = internal energy, W = work done, Q = Heat supplied then ______.
