Advertisements
Advertisements
प्रश्न
In an isochoric process, we have ____________.
विकल्प
W = 0
Q = 0
∆U = 0
∆T = 0
उत्तर
In an isochoric process, we have W = 0.
APPEARS IN
संबंधित प्रश्न
State the assumptions made for thermodynamic processes.
Write a note on free expansion.
Explain the thermodynamics of the isochoric process.
What is meant by a reversible and irreversible processes?
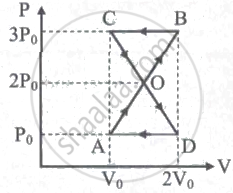
A thermodynamic system undergoes cyclic process ABCDA as shown in the figure. The work done by the system is ______
One mole of an ideal gas with `gamma` = 1.4 is adiabatically compressed so that its temperature rises from 27° C to 47° C. The change in the internal energy of the gas is (R = 8.3 J/mol.K) ____________.
Two identical samples of a gas are allowed to expand (i) isothermally (ii) adiabatically. Work done is ____________.
For an isothermal expansion of a perfect gas, the value of `(Delta "P")/"P"` is equal to ____________.
In an isothermal process, the volume of an ideal gas is halved. One can say that ____________.
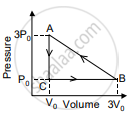
In the figure shown here, the work done in the process ACBA is ______.