Advertisements
Advertisements
प्रश्न
The efficiency of a heat engine working between the freezing point and boiling point of water is ____________.
विकल्प
6.25%
20%
26.8%
12.5%
उत्तर
The efficiency of a heat engine working between the freezing point and boiling point of water is 26.8%.
APPEARS IN
संबंधित प्रश्न
A gas contained in a cylinder surrounded by a thick layer of insulating material is quickly compressed has there been a transfer of heat?
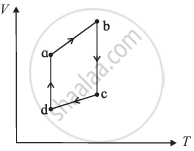
The figure shows the V-T diagram for one cycle of a hypothetical heat engine which uses the ideal gas. Draw the p-V diagram diagram of the system.
State Clausius form of the second law of thermodynamics.
State the second law of thermodynamics in terms of entropy.
Why does heat flow from a hot object to a cold object?
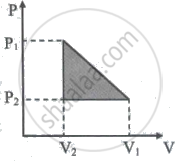
10 One mole of a van der Waals' gas obeying the equation `("P" + "a"/"V"^2)`(V - b) = RT undergoes the quasi-static cyclic process which is shown in the P-V diagram. The net heat absorbed by the gas in this process is ______
For a heat engine operating between temperatures t1 °C and t2 °C, its efficiency will be ______.
Let η1 is the efficiency of an engine at T1 = 447°C and T2 = 147°C while η2, is the efficiency at T1 = 947°C and T2 = 47°C. The ratio `eta_1/eta_2` will be ______.
What does a heat engine consist of?
Mention any two elements of a heat engine.
