Advertisements
Advertisements
प्रश्न
Give at least one example in case to show the structure of isomers of double bond compounds.
उत्तर १
In pentene
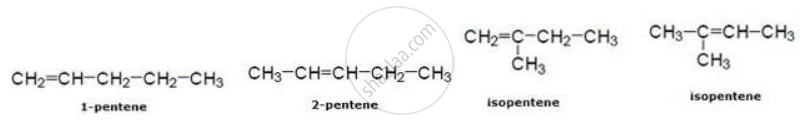
उत्तर २
\[\begin{array}{cc}
\ce{H}\phantom{.............}\ce{H}\phantom{....}\ce{H}\phantom{.}\\
|\phantom{...............}|\phantom{.....}|\phantom{.}\\
\ce{H - C^1 - C^2 = C^3 - C^4 - C^5 - H}\\
|\phantom{.....}|\phantom{.....}|\phantom{.....}|\phantom{.....}|\phantom{.}\\
\ce{H}\phantom{....}\ce{H}\phantom{....}|\phantom{.....}\ce{H}\phantom{....}\ce{H}\\
\ce{H - C - H}\phantom{..}\\
|\phantom{..}\\
\ce{H}\phantom{0}\\
\end{array}\]
3-methyl pent- 2-ene
APPEARS IN
संबंधित प्रश्न
Identify the term or substance based on the descriptions given below:
Hydrocarbon containing a triple bond used for welding purposes.
Give an example of isomers shown by triple bond hydrocarbons (alkynes) and write their IUPAC names.
Complete and balance the following equation. State the condition wherever necessary.
C2H4 + HCl → ______
Name a compound and draw the figure:
Cyclic compound with the triple bond.
Give the name of one member of the saturated hydrocarbons.
Give the name of one member of the unsaturated hydrocarbons.
Give the correct IUPAC name and the functional group for the compound whose structural formula is given below:
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{O}\\
\phantom{.}|\phantom{....}|\phantom{....}||\\
\ce{H - C - C - C - H}\\
|\phantom{....}|\phantom{.....}\\
\ce{H}\phantom{...}\ce{H}\phantom{.....}\\
\end{array}\]
Draw the structural formulae of the two isomers of Butane. Give the correct IUPAC name of each of the isomer.
Give one word for the following statement:
Organic compounds having the same molecular formula but different Structural formula.
The structures of four hydrocarbons are shown below:
| \[\begin{array}{cc} \phantom{.....}\ce{CH3}\\ \phantom{...}|\\ \ce{H3C - C - H}\\ \phantom{...}|\\ \phantom{.....}\ce{CH3} \end{array}\] |
\[\begin{array}{cc} \ce{CH3}\phantom{.....}\\ |\phantom{.......}\\ \ce{C = CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
\[\begin{array}{cc} \ce{H}\phantom{.....}\\ |\phantom{.....}\\ \ce{H3C - C - C - CH2}\\ |\phantom{....}|\\ \ce{H\phantom{...}H} \end{array}\] |
\[\begin{array}{cc} \phantom{...}\ce{CH3}\\ |\\ \ce{H3C - C = CH2} \end{array}\] |
How many isomers of butene are there?
