Advertisements
Advertisements
प्रश्न
Give at least one example in case to show the structure of isomers of double bond compounds.
उत्तर १
In pentene
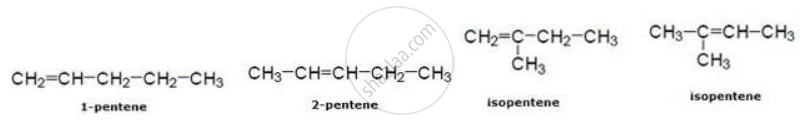
उत्तर २
\[\begin{array}{cc}
\ce{H}\phantom{.............}\ce{H}\phantom{....}\ce{H}\phantom{.}\\
|\phantom{...............}|\phantom{.....}|\phantom{.}\\
\ce{H - C^1 - C^2 = C^3 - C^4 - C^5 - H}\\
|\phantom{.....}|\phantom{.....}|\phantom{.....}|\phantom{.....}|\phantom{.}\\
\ce{H}\phantom{....}\ce{H}\phantom{....}|\phantom{.....}\ce{H}\phantom{....}\ce{H}\\
\ce{H - C - H}\phantom{..}\\
|\phantom{..}\\
\ce{H}\phantom{0}\\
\end{array}\]
3-methyl pent- 2-ene
APPEARS IN
संबंधित प्रश्न
Give the structural formulae of 2-methylpropane.
Give the structural formulae of Ethanoic acid.
Give balanced chemical equations for the ethanoic acid to ethyl ethanoate.
Complete and balance the following equation. State the condition wherever necessary.
C2H2 + Br2 → ______
Name a compound and draw the figure:
Cyclic compound with a single bond.
Name a compound and draw the figure:
Cyclic compound with the triple bond.
Give the name of one member of the saturated hydrocarbons.
Draw the structural formula for the following compound: diethyl ether
Name the following:
Organic compounds with same molecular formula but different structural formula.
The structures of four hydrocarbons are shown below:
| \[\begin{array}{cc} \phantom{.....}\ce{CH3}\\ \phantom{...}|\\ \ce{H3C - C - H}\\ \phantom{...}|\\ \phantom{.....}\ce{CH3} \end{array}\] |
\[\begin{array}{cc} \ce{CH3}\phantom{.....}\\ |\phantom{.......}\\ \ce{C = CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
\[\begin{array}{cc} \ce{H}\phantom{.....}\\ |\phantom{.....}\\ \ce{H3C - C - C - CH2}\\ |\phantom{....}|\\ \ce{H\phantom{...}H} \end{array}\] |
\[\begin{array}{cc} \phantom{...}\ce{CH3}\\ |\\ \ce{H3C - C = CH2} \end{array}\] |
How many isomers of butene are there?
