Advertisements
Advertisements
Question
Give at least one example in case to show the structure of isomers of double bond compounds.
Solution 1
In pentene
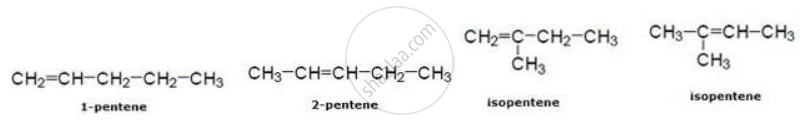
Solution 2
\[\begin{array}{cc}
\ce{H}\phantom{.............}\ce{H}\phantom{....}\ce{H}\phantom{.}\\
|\phantom{...............}|\phantom{.....}|\phantom{.}\\
\ce{H - C^1 - C^2 = C^3 - C^4 - C^5 - H}\\
|\phantom{.....}|\phantom{.....}|\phantom{.....}|\phantom{.....}|\phantom{.}\\
\ce{H}\phantom{....}\ce{H}\phantom{....}|\phantom{.....}\ce{H}\phantom{....}\ce{H}\\
\ce{H - C - H}\phantom{..}\\
|\phantom{..}\\
\ce{H}\phantom{0}\\
\end{array}\]
3-methyl pent- 2-ene
APPEARS IN
RELATED QUESTIONS
Identify the term or substance based on the descriptions given below:
Hydrocarbon containing a triple bond used for welding purposes.
Give the structural formulae of 2-methylpropane.
Complete and balance the following equation. State the condition wherever necessary.
CaC2 + H2O → ______
Complete and balance the following equation. State the condition wherever necessary.
\[\ce{C2H5OH ->[O][K2Cr2O7]}\] ______
What is the special feature of the structure of (i) ethene (ii) ethyne
Explain the following : Catenation
Give at least one example in case to show the structure of isomers of single-bond compounds.
Give at least one example in case to show the structure of isomers of a triple bond compound.
Give the correct IUPAC name and the functional group for the compound whose structural formula is given below:
\[\begin{array}{cc}
\ce{H\phantom{...}H\phantom{...}H\phantom{..}}\\
|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H - C - C - C - OH}\\
|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H\phantom{...}H\phantom{...}H\phantom{..}}
\end{array}\]
Name the following:
Organic compounds with same molecular formula but different structural formula.
