Advertisements
Advertisements
प्रश्न
Give one example in the case where supplying energy [given below] is necessary for a chemical reaction.
Catalyst
उत्तर
Catalyst:
It is a substance which alters the rate of the reaction [i.e. increases or decreases the rate] without taking part in the reaction.It itself remains chemically unchanged at the end of the reaction.
Example: Catalytic oxidation of ammonia – using catalyst platinum
\[\ce{4NH3 + 5O2 ->[Pt][800°C] 4NO + 6H2O + \triangle}\]
APPEARS IN
संबंधित प्रश्न
Balance the given chemical equation:
C + CO2 → CO
Write down what you understood from the following chemical reaction.
AgNO3(aq) + NaCI(aq) → AgCI ↓ + NaNO3(aq)
Write the balanced chemical equation of the following reaction. iron + sulphuric acid → ferrous sulphate + hydrogen.
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate mass of MnO2 used.
(a) Give only one suitable chemical test to identify the following gases.
- Ammonia
- Sulphur dioxide
- Hydrogen Chloride
- Chlorine
- Carbon Dioxide
- Oxygen
- Hydrogen
(b) Select a basic gas mentioned in Q.1 (a). How is the basic nature suspected?
(c)Select acidic gases from the gases mentioned in Q.1 (a). How is the acidic nature suspected?
(d) State the gas responsible for bleaching action.
(e)Which gas turn blue cobalt chloride paper light pink?
Write word equation for the following molecular equation:
\[\ce{NH3 [g] + HCl [g] ⇌ NH4Cl [s]}\]
Word equation:
State which characteristic is observed in the above reaction.
Give an-example of a similar above characteristic, seen between two other gaseous reactants.
Write word equation for the following molecular equation:
\[\ce{N2 + 3H2 ⇌[Fe - 450°C][200 atoms] 2NH3 + \triangle}\]
Word equation:
State why [+ Δ] indication is seen after NH3.
State what 200 atmospheres – indicates.
State the function of Fe in the above reaction.
Balance the following simple equation:
Al + O2 → Al2O3
Balance the following simple equation:
K + H2O → KOH + H2
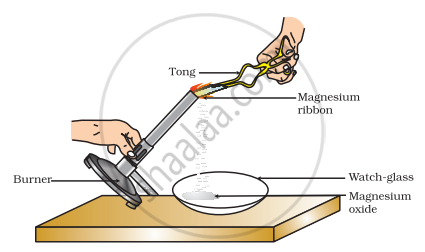
Which of the following is the correct observation of the reaction shown in the above set up?
