Advertisements
Advertisements
प्रश्न
Give reason:
Sodium, potassium and lithium are stored under oil.
उत्तर
- All three metals react with water, producing a lot of heat. As a result, hydrogen evolves and catches fire.
- They cannot be kept in the air because air contains moisture or water vapours. They are kept under kerosene to avoid contact with both air and water.
संबंधित प्रश्न
Give two examples of amphoteric oxides.
Name two metals which will displace hydrogen from dilute acids, and two metals which will not.
What are amphoteric oxides?
Diamond : electric insulator : : _______ : electric conductor
Which of the following oxide(s) of iron would be obtained on prolonged reaction of iron with steam?
Silver articles become black on prolonged exposure to air. This is due to the formation of
Stainless steel is very useful material for our life. In stainless steel, iron is mixed with
If copper is kept open in air, it slowly loses its shining brown surface and gains a green coating. It is due to the formation of
When a metal X is treated with cold water, it gives a basic salt Y with molecular formula XOH (Molecular mass = 40) and liberates a gas Z which easily catches fire. Identify X, Y and Z and also write the reaction involved.
Which of the following reacts with cold water vigorously?
Zinc sulphate forms a colourless solution in water. Will you observe any colour on adding copper turning in it?
Out of three metals, sodium, calcium and aluminium, which metal reacts most with water? Give the equation of reaction with all three metals.
Answer the questions based on the figure below:
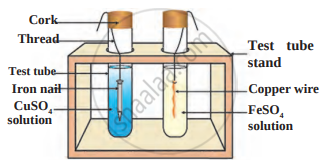
- Which experiment setup is demonstrated in the figure?
- What do you conclude after the reactions? Name reaction.
Answer the questions based on the figure below:
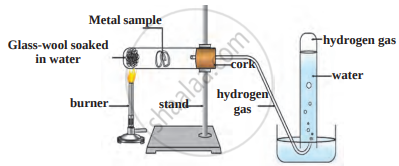
- Which reaction is shown in the figure?
- Which gas is evolved in the reaction?
- Give an example of reactants that rapidly show this reaction. Give equations.
- Give an example of reactants that do not react rapidly.
- In what condition will reactants of (c) part react? Give equation.
Metal oxides generally react with acids, but few oxides of metal also react with bases. Such metallic oxides are:
- MgO
- ZnO
- Al2O3
- Cao
On adding dilute sulphuric acid to a test tube containing a metal ‘X’, a colourless gas is produced when a burning match stick is brought near it. Which of the following correctly represents the metal ‘X’?
