Advertisements
Advertisements
प्रश्न
Give the equation of state for an adiabatic process.
उत्तर
The equation of state for an adiabatic process is given by PVγ = Constant
Here γ is called adiabatic exponent `(γ = "C"_"p"/"C"_γ)` which depends on the nature of the gas
APPEARS IN
संबंधित प्रश्न
Explain the cyclic process.
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
Explain work done during a thermodynamic process.
When a cycle tyre suddenly bursts, the air inside the tyre expands. This process is ____________.
In an isochoric process, we have ____________.
Explain in detail an adiabatic process.
Draw the TP diagram (P-x axis, T-y axis), VT(T-x axis, V-y axis) diagram for
- Isochoric process
- Isothermal process
- Isobaric process
Ideal gas for which 'ϒ' = 1.5 is suddenly compressed to `1/4`th of its initial volume. The ratio of 4 the final pressure to the initial pressure is ______.
`(ϒ = "C"_"p"/"C"_"v")`
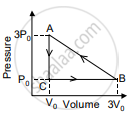
In the figure shown here, the work done in the process ACBA is ______.
In a cyclic process, if ΔU = internal energy, W = work done, Q = Heat supplied then ______.
