Advertisements
Advertisements
प्रश्न
Give the mathematical expression that relates gas volume and moles.
उत्तर
- The mathematical relationship betwêen the volume of a gas and the number of moles is V α n
- `"V"_1/"n"_1 = "V"_2/"n"_2` = Constant
Where V1 and n1 are the volume and number of moles of a gas and V2 and n2 are the values of volume and number of moles of the same gas at a different set of conditions. - If the volume of the gas increase then the number of moles of the gas also increases.
- At a certain temperature and pressure, the volume of a gas is directly proportional to the number of moles of the gas.
APPEARS IN
संबंधित प्रश्न
Give reason for the following:
Gases have a lower density compared to solids or liquids.
What is meant by aqueous tension? How is the pressure exerted by a gas corrected to account for aqueous tension?
Convert the following temperature from degree Celcius to kelvin.
25° C
Convert 89 kPa to newton per square metre (Nm−2)
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
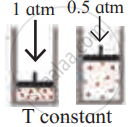 |
______________ |
Consider a sample of a gas in a cylinder with a movable piston.

Show diagrammatically the changes in the position of the piston, if the temperature is decreased from 400 K to 300 K, and pressure is decreased from 4 bar to 3 bar.
Solve the following.
A balloon is inflated with helium gas at room temperature of 25°C and at 1 bar pressure when its initial volume is 2.27L and allowed to rise in the air. As it rises in the air external pressure decreases and the volume of the gas increases till finally, it bursts when external pressure is 0.3bar. What is the limit at which the volume of the balloon can stay inflated?
A sample of gas at 15°C at 1 atm. has a volume of 2.58 dm3. When the temperature is raised to 38°C at 1 atm does the volume of the gas Increase? If so, calculate the final volume.
Volume of a balloon at 25°C and 1 bar pressure is 2.27 L. If the pressure of the gas in balloon is reduced to 0.227 bar, what is the rise in volume of a gas?
If 300 mL of a gas at 26.85°C is cooled to 6.85°C at constant pressure. What will be the final volume of gas?
