Advertisements
Advertisements
प्रश्न
Give the mathematical expression that relates gas volume and moles.
उत्तर
- The mathematical relationship betwêen the volume of a gas and the number of moles is V α n
- `"V"_1/"n"_1 = "V"_2/"n"_2` = Constant
Where V1 and n1 are the volume and number of moles of a gas and V2 and n2 are the values of volume and number of moles of the same gas at a different set of conditions. - If the volume of the gas increase then the number of moles of the gas also increases.
- At a certain temperature and pressure, the volume of a gas is directly proportional to the number of moles of the gas.
APPEARS IN
संबंधित प्रश्न
Convert the following temperature from degree Celcius to kelvin.
−197° C
Convert exactly 1.5 atm to pascals
Convert 101.325 kPa to bar.
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
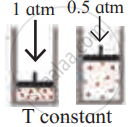 |
______________ |
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
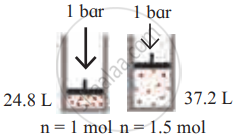 |
______________ |
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called __________.
Explain the following observation.
The type of an automobile is inflated to slightly lesser pressure in summer than in winter
Explain the following observation.
The size of a weather balloon becomes larger and larger as it ascends up to larger altitude
Sulphur hexafluoride is a colourless, odourless gas; calculate the pressure exerted by 1.82 moles of the gas in a steel vessel of volume 5.43 dm3 at 69.5 °C, assuming ideal gas behaviour
A gas occupies a volume of 4.2 dm3 at 101 kPa pressure. What volume will gas occupy if the pressure is increased to 235 kPa keeping the temperature constant?
