Advertisements
Advertisements
प्रश्न
Convert exactly 1.5 atm to pascals
उत्तर
Exactly 1.5 atm to pascals:
1 atm = 101325 Pa
∴ 1.5 atm = 1.5 × 101325
= 151987.5 Pa
APPEARS IN
संबंधित प्रश्न
Explain Why?
"When stating the volume of a gas, the pressure and temperature should also be given."
State (i) the three variables for gas laws and (ii) SI units of these variables.
Give reason for the following:
Gases exert pressure in all directions.
What is meant by aqueous tension? How is the pressure exerted by a gas corrected to account for aqueous tension?
State the following:
The absolute temperature of a gas at 7°C
Convert the following temperature from degree Celcius to kelvin.
273° C
Convert the following pressure value into Pascals.
1 atmosphere
Convert −100° C to kelvin
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
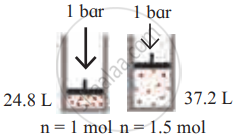 |
______________ |
Match the pairs of the following:
| Column ‘A’ | Column ‘B’ |
| a. Boyle’s law | i. at constant pressure and volume |
| b. Charles’ law | ii. at constant temperature |
| iii. at constant pressure |
With the help of the graph answer the following -

At constant temperature, Identify the law.
With the help of the graph answer the following -

At constant temperature, Write the statement of law.
Solve the following.
A syringe has a volume of 10.0 cm3 at pressure 1 atm. If you plug the end so that no gas can escape and push the plunger down, what must be the final volume to change the pressure to 3.5 atm?

Solve the following.
A hot air balloon has a volume of 2800 m3 at 99°C. What is the volume if the air cools to 80°C?

The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called __________.
Give the mathematical expression that relates gas volume and moles.
Explain the following observation.
Aerated water bottles are kept under water during summer
Explain the following observation.
The type of an automobile is inflated to slightly lesser pressure in summer than in winter
A small bubble rises from the bottom of a lake where the temperature and pressure are 6°C and 4 atm. to the water surface, where the temperature is 25°C and pressure is 1 atm. Calculate the final volume in (mL) of the bubble, if its initial volume is 1.5 mL.
For a given mass of an ideal gas, which of the following statements is CORRECT?
Volume of a balloon at 25°C and 1 bar pressure is 2.27 L. If the pressure of the gas in balloon is reduced to 0.227 bar, what is the rise in volume of a gas?
A certain mass of a gas occupies a volume of 2 dm3 at STP. At what temperature the volume of gas becomes double, keeping the pressure constant?
At what temperature the volume of a gas becomes absolutely zero?
The volume of 400 cm3 chlorine gas at 400 mm of Hg is decreased to 200 cm3 at constant temperature. What is the new pressure of gas?
10 g of gas at one atomospheric pressure is cooled from 273.15°C to 0°C keeping the volume constant. What is the final pressure?
The number of molecules in 8.96 litres of gas at 0°C and 1 atm. pressure is approximately ______.
