Advertisements
Advertisements
प्रश्न
What is meant by aqueous tension? How is the pressure exerted by a gas corrected to account for aqueous tension?
उत्तर
Gases such as nitrogen and hydrogen are collected over water as shown in the diagram. When the gas is collected over water, the gas is moist and contains water vapour. The total pressure exerted by this moist gas is equal to the sum of the partial pressures of the dry gas and the pressure exerted by water vapour. The partial pressure of water vapour is also known as aqueous tension.

Ptotal = Pgas + Pwater vapour
Pgas = Ptotal- Pwater vapour
Actual pressure of gas = Total pressure - Aqueous tension
APPEARS IN
संबंधित प्रश्न
What would be the mass of CO2 occupying a volume of 44 litres at 25°C and 750 mm pressure.
Convert the following pressure value into Pascals.
107000 Nm−2
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
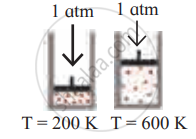 |
______________ |
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
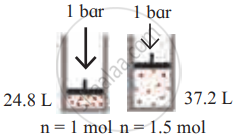 |
______________ |
With the help of the graph answer the following -

At constant temperature, the Graph shows the relationship between pressure and volume. Represent the relation mathematically.
Solve the following.
A balloon is inflated with helium gas at room temperature of 25°C and at 1 bar pressure when its initial volume is 2.27L and allowed to rise in the air. As it rises in the air external pressure decreases and the volume of the gas increases till finally, it bursts when external pressure is 0.3bar. What is the limit at which the volume of the balloon can stay inflated?
Give the mathematical expression that relates gas volume and moles.
A sample of gas has a volume of 8.5 dm3 at an unknown temperature. When the sample is submerged in ice water at 0°C, its volume gets reduced to 6.37 dm3. What is its initial temperature?
Of two samples of nitrogen gas, sample A contains 1.5 moles of nitrogen in a vessel of the volume of 37.6 dm3 at 298 K, and sample B is in a vessel of volume 16.5 dm3 at 298 K. Calculate the number of moles in sample B.
According to Andrews isothermals at what temperature the carbon dioxide gas starts to condense at 73 atmosphere?
