Advertisements
Advertisements
प्रश्न
How did Charles's law lead to the concept of an absolute scale of temperature?
उत्तर
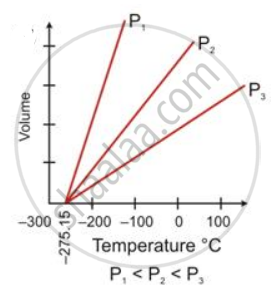
The temperature scale with its zero at -273°C and where each degree is equal to the degree on the Celsius scale is called the absolute scale of temperature.
The temperature of -273°C is called absolute zero. Theoretically, this is the lowest temperature which can never be reached. All molecular motion ceases at this temperature.
The temperature of -273°C is called absolute zero.
V = V° `((273 + "t")/273)`
Volume at - 273°C = V° `((273 - 273)/273) = 0`
APPEARS IN
संबंधित प्रश्न
State the law which is represented by the following graph:
Choose the correct answer:
The absolute temperature value that corresponds to 27°C is
What will be the minimum pressure required to compress 500 dm3 of air at 1 bar to 200 dm3 temperature remaining constant?
2 liters of gas is enclosed in a vessel at a pressure of 760 mmHg. If the temperature remains constant, calculate pressure when volume changes to 4 dm3.
Calculate the final volume of a gas 'X' if the original pressure of the gas at STP is doubled and its temperature is increased three times.
It is found that on heating a gas its volume increases by 50% and its pressure decreases to 60% of its original value. If the original temperature was -15°C, find the temperature to which it was heated.
2500 cm3 of hydrogen is taken at STP. The pressure of this gas is further increased by two and a half times (temperature remaining constant). What volume will hydrogen occupy now?
A certain mass of gas occupied 850 ml at a pressure of 760 mm of Hg. On increasing the pressure it was found that the volume of the gas was 75% of its initial value. Assuming constant temperature, find the final pressure of the gas?
Fill in the blank with the correct word, from the words in option:
If the temperature of a fixed mass of a gas is kept constant and the pressure is increased, the volume correspondingly _______.
A fixed volume of a gas occupies 760cm3 at 27°C and 70 cm of Hg. What will be its vol. at s.t.p.
