Advertisements
Advertisements
प्रश्न
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
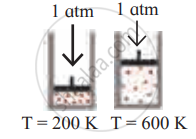 |
______________ |
उत्तर
| Diagram | Gas laws |
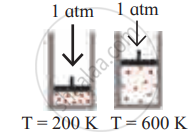 |
Charles’ law |
APPEARS IN
संबंधित प्रश्न
Explain Why?
"When stating the volume of a gas, the pressure and temperature should also be given."
What would be the mass of CO2 occupying a volume of 44 litres at 25°C and 750 mm pressure.
What is meant by aqueous tension? How is the pressure exerted by a gas corrected to account for aqueous tension?
Answer in one sentence.
A bubble of methane gas rises from the bottom of the North sea. What will happen to the size of the bubble as it rises to the surface?
Convert the following pressure value into Pascals.
107000 Nm−2
Convert exactly 1.5 atm to pascals
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
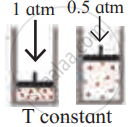 |
______________ |
Consider a sample of a gas in a cylinder with a movable piston.

Show diagrammatically the changes in the position of the piston, if the temperature is decreased from 300 K to 150 K at constant pressure.
With the help of the graph answer the following -

At constant temperature, Write the statement of law.
Solve the following.
A balloon is inflated with helium gas at room temperature of 25°C and at 1 bar pressure when its initial volume is 2.27L and allowed to rise in the air. As it rises in the air external pressure decreases and the volume of the gas increases till finally, it bursts when external pressure is 0.3bar. What is the limit at which the volume of the balloon can stay inflated?
Solve the following.
The volume of a given mass of a gas at 0°C is 2 dm3. Calculate the new volume of the gas at constant pressure when the temperature is increased by 10°C.
Solve the following.
At 0°C, a gas occupies 22.4 liters. How much hot must be the gas in celsius and in kelvin to reach a volume of 25.0 liters?
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called __________.
State Boyle's law.
Name two items that can serve as a model for Gay Lusaac’s law and explain.
Explain the following observation.
Aerated water bottles are kept under water during summer
Explain the following observation.
The type of an automobile is inflated to slightly lesser pressure in summer than in winter
Explain the following observation.
The size of a weather balloon becomes larger and larger as it ascends up to larger altitude
A sample of gas at 15°C at 1 atm. has a volume of 2.58 dm3. When the temperature is raised to 38°C at 1 atm does the volume of the gas Increase? If so, calculate the final volume.
A sample of gas has a volume of 8.5 dm3 at an unknown temperature. When the sample is submerged in ice water at 0°C, its volume gets reduced to 6.37 dm3. What is its initial temperature?
Of two samples of nitrogen gas, sample A contains 1.5 moles of nitrogen in a vessel of the volume of 37.6 dm3 at 298 K, and sample B is in a vessel of volume 16.5 dm3 at 298 K. Calculate the number of moles in sample B.
For a given mass of an ideal gas, which of the following statements is CORRECT?
A certain sample of gas has a volume of 0.2 L at one atmosphere pressure and 273.15 K. What is the volume of gas at 273.15°C at same pressure?
At what temperature the volume of a gas becomes absolutely zero?
If 2 moles of an ideal gas at 546 K has volume of 44.8 L, then what will be it's pressure? (R = 0.082)
10 g of gas at one atomospheric pressure is cooled from 273.15°C to 0°C keeping the volume constant. What is the final pressure?
