Advertisements
Advertisements
प्रश्न
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
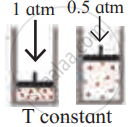 |
______________ |
उत्तर
| Diagram | Gas laws |
 |
Boyle’s law |
APPEARS IN
संबंधित प्रश्न
Explain Why?
"When stating the volume of a gas, the pressure and temperature should also be given."
State (i) the three variables for gas laws and (ii) SI units of these variables.
Give reason for the following:
Gases have a lower density compared to solids or liquids.
Convert the following temperature from degree Celcius to kelvin.
273° C
Convert the following pressure value into Pascals.
10 atmosphere
Convert 0.124 torr to the standard atmosphere
Consider a sample of a gas in a cylinder with a movable piston.

Show diagrammatically the changes in the position of the piston, if the temperature is decreased from 400 K to 300 K, and pressure is decreased from 4 bar to 3 bar.
Match the pairs of the following:
| Column ‘A’ | Column ‘B’ |
| a. Boyle’s law | i. at constant pressure and volume |
| b. Charles’ law | ii. at constant temperature |
| iii. at constant pressure |
Write the statement for Boyle’s law
With the help of the graph answer the following -

At constant temperature, the Graph shows the relationship between pressure and volume. Represent the relation mathematically.
Solve the following.
The volume of a given mass of a gas at 0°C is 2 dm3. Calculate the new volume of the gas at constant pressure when the temperature is increased by 10°C.
Assertion: Critical temperature of CO2 is 304 K, it can be liquefied above 304 K.
Reason: For a given mass of gas, volume is to directly proportional to pressure at constant temperature
Give the mathematical expression that relates gas volume and moles.
Explain the following observation.
The size of a weather balloon becomes larger and larger as it ascends up to larger altitude
A sample of gas has a volume of 8.5 dm3 at an unknown temperature. When the sample is submerged in ice water at 0°C, its volume gets reduced to 6.37 dm3. What is its initial temperature?
Sulphur hexafluoride is a colourless, odourless gas; calculate the pressure exerted by 1.82 moles of the gas in a steel vessel of volume 5.43 dm3 at 69.5 °C, assuming ideal gas behaviour
A small bubble rises from the bottom of a lake where the temperature and pressure are 6°C and 4 atm. to the water surface, where the temperature is 25°C and pressure is 1 atm. Calculate the final volume in (mL) of the bubble, if its initial volume is 1.5 mL.
Hydrochloric acid is treated with a metal to produce hydrogen gas. Suppose a student carries out this reaction and collects a volume of 154.4 × 10−3 dm3 of a gas at a pressure of 742 mm of Hg at a temperature of 298 K. What mass of hydrogen gas (in mg) did the student collect?
For a given mass of an ideal gas, which of the following statements is CORRECT?
According to Andrews isothermals, the minimum temperature at which carbon dioxide gas obeys Boyles law is ______.
A certain mass of a gas occupies a volume of 2 dm3 at STP. At what temperature the volume of gas becomes double, keeping the pressure constant?
If 300 mL of a gas at 26.85°C is cooled to 6.85°C at constant pressure. What will be the final volume of gas?
The number of molecules in 8.96 litres of gas at 0°C and 1 atm. pressure is approximately ______.
