Advertisements
Advertisements
प्रश्न
State (i) the three variables for gas laws and (ii) SI units of these variables.
उत्तर
- Three variables for gas laws: Volume (V), Pressure (P), Temperature (T)
- SI units of these variables:
For volume: Cubic metre (m3)
For pressure: Pascal (Pa)
For temperature: Kelvin (K)
APPEARS IN
संबंधित प्रश्न
Explain Why?
"When stating the volume of a gas, the pressure and temperature should also be given."
Answer in one sentence.
A bubble of methane gas rises from the bottom of the North sea. What will happen to the size of the bubble as it rises to the surface?
Convert the following pressure value into Pascals.
1 kPa
Convert the following pressure value into Pascals.
107000 Nm−2
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
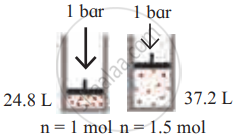 |
______________ |
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called __________.
Explain the following observation.
Aerated water bottles are kept under water during summer
A certain sample of gas has a volume of 0.2 L at one atmosphere pressure and 273.15 K. What is the volume of gas at 273.15°C at same pressure?
According to Andrews isothermals, the minimum temperature at which carbon dioxide gas obeys Boyles law is ______.
A certain mass of a gas occupies a volume of 2 dm3 at STP. At what temperature the volume of gas becomes double, keeping the pressure constant?
