Advertisements
Advertisements
प्रश्न
State Boyle's Law.
उत्तर १
Robert Boyle systematically studied the relationship between pressure and volume of gases. In 1962, he found that, at a constant temperature, the volume of a fixed mass of a dry gas decreased by half when the pressure on it was doubled, and it became four times its original volume when its pressure was decreased to on-fourth. He described this behaviour in the form of a law, known as Boyle's Law.
उत्तर २
Boyle’s Law : “Temperature remaining constant the volume of a given mass of dry gas is inversely proportional to its pressure.”
V α `1/"P"` = T = Constant
APPEARS IN
संबंधित प्रश्न
Explain Why?
Inflating a balloon seems to violate Boyle's law.
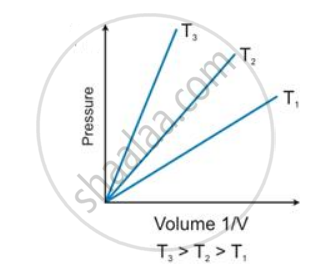
State the law which is represented by the following graph:
561 dm3 of a gas at STP is filled in a 748 dm3 container. If the temperature is constant, calculate the percentage change in pressure required.
A certain amount of a gas occupies a volume of 0.4 litre at 17°C. To what temperature should it be heated so that its volume gets (a) doubled, (b) reduced to half, pressure remaining constant?
A certain mass of a gas occupies 2 litres at 27°C and 100 Pa. Find the temperature when volume and pressure become half of their initial values.
Correct the following statement:
The volume of a gas is inversely proportional to its pressure at a constant temperature.
50 cm3 of hydrogen is collected over water at 17°C and 750 mmHg pressure. Calculate the volume of a dry gas at STP. The water vapour pressure at 17°C is 14 mmHg.
Name or state the following:
The law which studies the relationship between pressure of a gas and the volume occupied by it at constant temperature.
Fill in the blank with the correct word, from the words in option:
1 dm3 of a gas is equal to _______.
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is _______.
