Advertisements
Advertisements
प्रश्न
Explain the following observation.
The size of a weather balloon becomes larger and larger as it ascends up to larger altitude
उत्तर
The volume of the gas is inversely proportional to the pressure at a given temperature according to Boyle’s law. As the weather balloon ascends, the pressure tends to decrease. As a result, the volume of the gas inside the balloon or the size of the balloon is likely to increase.
APPEARS IN
संबंधित प्रश्न
What would be the mass of CO2 occupying a volume of 44 litres at 25°C and 750 mm pressure.
State (i) the three variables for gas laws and (ii) SI units of these variables.
Convert the following pressure value into Pascals.
10 atmosphere
Convert the following pressure value into Pascals.
1 kPa
Hot air balloons float in the air because of the low density of the air inside the balloon. Explain this with the help of an appropriate gas law.

Identify the gas laws from the following diagram.
| Diagram | Gas laws |
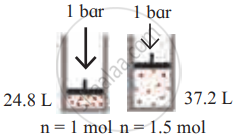 |
______________ |
Solve the following.
A syringe has a volume of 10.0 cm3 at pressure 1 atm. If you plug the end so that no gas can escape and push the plunger down, what must be the final volume to change the pressure to 3.5 atm?

Explain the following observation.
Aerated water bottles are kept under water during summer
Explain the following observation.
Liquid ammonia bottle is cooled before opening the seal
Isochor is the graph plotted between ______.
