Advertisements
Advertisements
Question
Explain the following observation.
The size of a weather balloon becomes larger and larger as it ascends up to larger altitude
Solution
The volume of the gas is inversely proportional to the pressure at a given temperature according to Boyle’s law. As the weather balloon ascends, the pressure tends to decrease. As a result, the volume of the gas inside the balloon or the size of the balloon is likely to increase.
APPEARS IN
RELATED QUESTIONS
Convert the following temperature from degree Celcius to kelvin.
−15° C
Convert the following pressure value into Pascals.
107000 Nm−2
Convert exactly 1.5 atm to pascals
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
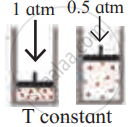 |
______________ |
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
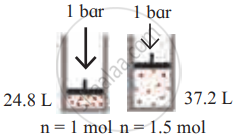 |
______________ |
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called __________.
State Boyle's law.
Name two items that can serve as a model for Gay Lusaac’s law and explain.
Of two samples of nitrogen gas, sample A contains 1.5 moles of nitrogen in a vessel of the volume of 37.6 dm3 at 298 K, and sample B is in a vessel of volume 16.5 dm3 at 298 K. Calculate the number of moles in sample B.
At what temperature, the volume of gas would become zero?
