Advertisements
Advertisements
Question
Of two samples of nitrogen gas, sample A contains 1.5 moles of nitrogen in a vessel of the volume of 37.6 dm3 at 298 K, and sample B is in a vessel of volume 16.5 dm3 at 298 K. Calculate the number of moles in sample B.
Solution
nA = 1.5 mol
nB = ?
VA = 37.6 dm3
VB = 16.5 dm3
(T = 298 K constant)
`"V"_"A"/"n"_"A" = "V"_"B"/"n"_"B"`
`37.6/1.5 = 16.5/"n"_"B"`
`25.06 = 16.5/"n"_"B"`
`"n"_"B" = 16.5/25.06`
`"n"_"B"` = 0.658 mol
∴ The number of moles in sample B is 0.658 mol
APPEARS IN
RELATED QUESTIONS
Convert the following temperature from degree Celcius to kelvin.
273° C
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
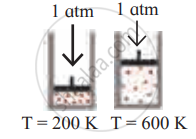 |
______________ |
Consider a sample of a gas in a cylinder with a movable piston.

Show diagrammatically the changes in the position of the piston, if the temperature is decreased from 400 K to 300 K, and pressure is decreased from 4 bar to 3 bar.
Explain the following observation.
The type of an automobile is inflated to slightly lesser pressure in summer than in winter
A sample of gas has a volume of 8.5 dm3 at an unknown temperature. When the sample is submerged in ice water at 0°C, its volume gets reduced to 6.37 dm3. What is its initial temperature?
Volume of a balloon at 25°C and 1 bar pressure is 2.27 L. If the pressure of the gas in balloon is reduced to 0.227 bar, what is the rise in volume of a gas?
At what temperature the volume of a gas becomes absolutely zero?
Isochor is the graph plotted between ______.
The volume of 400 cm3 chlorine gas at 400 mm of Hg is decreased to 200 cm3 at constant temperature. What is the new pressure of gas?
At what temperature, the volume of gas would become zero?
