Advertisements
Advertisements
प्रश्न
Of two samples of nitrogen gas, sample A contains 1.5 moles of nitrogen in a vessel of the volume of 37.6 dm3 at 298 K, and sample B is in a vessel of volume 16.5 dm3 at 298 K. Calculate the number of moles in sample B.
उत्तर
nA = 1.5 mol
nB = ?
VA = 37.6 dm3
VB = 16.5 dm3
(T = 298 K constant)
`"V"_"A"/"n"_"A" = "V"_"B"/"n"_"B"`
`37.6/1.5 = 16.5/"n"_"B"`
`25.06 = 16.5/"n"_"B"`
`"n"_"B" = 16.5/25.06`
`"n"_"B"` = 0.658 mol
∴ The number of moles in sample B is 0.658 mol
APPEARS IN
संबंधित प्रश्न
Give reason for the following:
Gases exert pressure in all directions.
State the following:
The absolute temperature of a gas at 7°C
Hot air balloons float in the air because of the low density of the air inside the balloon. Explain this with the help of an appropriate gas law.

Identify the gas laws from the following diagram.
| Diagram | Gas laws |
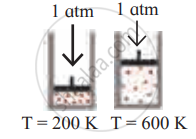 |
______________ |
Consider a sample of a gas in a cylinder with a movable piston.

Show diagrammatically the changes in the position of the piston, if the temperature is decreased from 400 K to 300 K, and pressure is decreased from 4 bar to 3 bar.
Solve the following.
A hot air balloon has a volume of 2800 m3 at 99°C. What is the volume if the air cools to 80°C?

Assertion: Critical temperature of CO2 is 304 K, it can be liquefied above 304 K.
Reason: For a given mass of gas, volume is to directly proportional to pressure at constant temperature
A certain sample of gas has a volume of 0.2 L at one atmosphere pressure and 273.15 K. What is the volume of gas at 273.15°C at same pressure?
Volume of a balloon at 25°C and 1 bar pressure is 2.27 L. If the pressure of the gas in balloon is reduced to 0.227 bar, what is the rise in volume of a gas?
At what temperature, the volume of gas would become zero?
