Advertisements
Advertisements
प्रश्न
Give reason for the following:
Gases have a lower density compared to solids or liquids.
उत्तर
Gases have low densities as the inter-molecular distance between the molecules of gases is very large.
Pressure on the wall = `("Force exerted on the wall")/("Total area of the container wall")`
संबंधित प्रश्न
What would be the mass of CO2 occupying a volume of 44 litres at 25°C and 750 mm pressure.
State the following:
The absolute temperature of a gas at 7°C
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
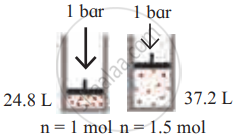 |
______________ |
Write the statement for Charles’ law
Solve the following.
A hot air balloon has a volume of 2800 m3 at 99°C. What is the volume if the air cools to 80°C?

Solve the following.
At 0°C, a gas occupies 22.4 liters. How much hot must be the gas in celsius and in kelvin to reach a volume of 25.0 liters?
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called __________.
At 25°C and 1 atm, a cylinder containing 10 L of an ideal gas is connected to the empty cylinder with a capacity of 20 L. The pressures exerted by gas m both the cylinders will be ____________.
According to Andrews isothermals, the minimum temperature at which carbon dioxide gas obeys Boyles law is ______.
Isochor is the graph plotted between ______.
