Advertisements
Advertisements
Question
Give reason for the following:
Gases have a lower density compared to solids or liquids.
Solution
Gases have low densities as the inter-molecular distance between the molecules of gases is very large.
Pressure on the wall = `("Force exerted on the wall")/("Total area of the container wall")`
RELATED QUESTIONS
Convert the following temperature from degree Celcius to kelvin.
273° C
Convert −100° C to kelvin
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
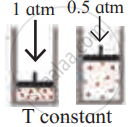 |
______________ |
With the help of the graph answer the following -

At constant temperature, Write the statement of law.
Solve the following.
The volume of a given mass of a gas at 0°C is 2 dm3. Calculate the new volume of the gas at constant pressure when the temperature is increased by 10°C.
State Boyle's law.
Explain the following observation.
Aerated water bottles are kept under water during summer
Explain the following observation.
Liquid ammonia bottle is cooled before opening the seal
A sample of gas at 15°C at 1 atm. has a volume of 2.58 dm3. When the temperature is raised to 38°C at 1 atm does the volume of the gas Increase? If so, calculate the final volume.
At what temperature, the volume of gas would become zero?
