Advertisements
Advertisements
Question
Explain the following observation.
Liquid ammonia bottle is cooled before opening the seal
Solution
Liquid ammonia bottle contains the gas under very high pressure. If the bottle is opened as such, then the sudden decrease in pressure will lead to a large increase in the volume of the gas. As a result, the gas will come out of the bottle all of a sudden with force. This will lead to the breakage of the bottle and also causes an accident.
However, if the bottle is cooled under tap water for some time, there will be a decrease in the volume of a gas to a large extent. if the seal is opened now, the gas will come out of the bottle at a slower rate, reduces the chances of an accident.
APPEARS IN
RELATED QUESTIONS
Convert the following pressure value into Pascals.
10 atmosphere
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
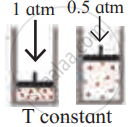 |
______________ |
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
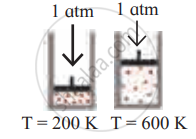 |
______________ |
With the help of the graph answer the following -

At constant temperature, the Graph shows the relationship between pressure and volume. Represent the relation mathematically.
With the help of the graph answer the following -

At constant temperature, Write the statement of law.
Solve the following.
The volume of a given mass of a gas at 0°C is 2 dm3. Calculate the new volume of the gas at constant pressure when the temperature is decreased by 10°C.
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called __________.
Explain the following observation.
Aerated water bottles are kept under water during summer
Explain the following observation.
The size of a weather balloon becomes larger and larger as it ascends up to larger altitude
Sulphur hexafluoride is a colourless, odourless gas; calculate the pressure exerted by 1.82 moles of the gas in a steel vessel of volume 5.43 dm3 at 69.5 °C, assuming ideal gas behaviour
