Advertisements
Advertisements
प्रश्न
Heavy stable nucle have more neutrons than protons. This is because of the fact that ______.
विकल्प
neutrons are heavier than protons.
electrostatic force between protons are repulsive.
neutrons decay into protons through beta decay.
nuclear forces between neutrons are weaker than that between protons.
उत्तर
Heavy stable nucle have more neutrons than protons. This is because of the fact that electrostatic force between protons are repulsive.
Explanation:
Neutron-proton ratio
- For lighter nuclei, the greatest stability is achieved when the number of protons and neutrons are approximately equal (N = Z), ie.
- Heavy nuclei are stable only when they have more neutrons than protons. Thus heavy nuclei are neutron-rich compared to lighter nuclei (for heavy nuclei, more the protons in the nucleus, the greater the electrical repulsive force between them. Therefore more neutrons are added to provide the strong attractive forces necessary to keep the nucleus stable.)
APPEARS IN
संबंधित प्रश्न
Write symbolically the nuclear β+ decay process of
The neutron separation energy is defined as the energy required to remove a neutron from the nucleus. Obtain the neutron separation energies of the nuclei
Define the terms (i) half-life (T1/2) and (ii) average life (τ). Find out their relationships with the decay constant (λ).
What is the minimum energy which a gamma-ray photon must possess in order to produce electron-positron pair?
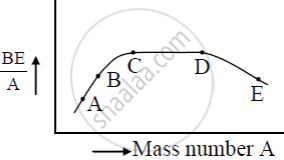
The figure shows the plot of binding energy (BE) per nucleon as a function of mass number A. The letters A, B, C, D, and E represent the positions of typical nuclei on the curve. Point out, giving reasons, the two processes (in terms of A, B, C, D, and E ), one of which can occur due to nuclear fission and the other due to nuclear fusion.
Determine the binding energy per nucleon of the americium isotope
The difference in mass of a nucleus and its constituents is called ______.
Calculate the binding energy of an alpha particle in MeV. Given
mass of a proton = 1.007825 u
mass of a neutron = 1.008665 u
mass of He nucleus = 4.002800 u
1u = 931 MeV/c2
Calculate the values of x and y in the following nuclear reaction.
What is binding energy of nucleus?
