Advertisements
Advertisements
प्रश्न
Highest oxidation state of manganese in fluoride is \[\ce{+4 (MnF4)}\] but highest oxidation state in oxides is \[\ce{+7 (Mn2O7)}\] because ______.
विकल्प
fluorine is more electronegative than oxygen.
fluorine does not possess d-orbitals.
fluorine stabilises lower oxidation state.
in covalent compounds fluorine can form single bond only while oxygen forms double bond.
उत्तर
Highest oxidation state of manganese in fluoride is \[\ce{+4 (MnF4)}\] but highest oxidation state in oxides is \[\ce{+7 (Mn2O7)}\] because in covalent compounds fluorine can form single bond only while oxygen forms double bond.
Explanation:
Oxygen has the capacity to form multiple bonds which enables it to form a variety of covalent compounds.
In \[\ce{(Mn2O7)}\] also, 6 oxygen are doubly bonded to two manganese atoms and one oxygen is forming bridge between two.
While in \[\ce{(MnF4)}\], four fluorine atoms are singly bonded to manganese atom giving it a +4 oxidation state.
Therefore, due to capability of oxygen to have multiple bonds in covalent compounds, manganese is having higher oxidation state of +7 in \[\ce{(Mn2O7)}\].
APPEARS IN
संबंधित प्रश्न
How would you account for the following?
Transition metals exhibit variable oxidation states.
Comment on the statement that elements of the first transition series possess many properties different from those of heavier transition elements.
An antifriction alloy made up of antimony with tin and copper, which is extensively used in machine bearings is called _______.
(A) Duralumin
(B) Babbitt metal
(C) Spiegeleisen
(D) Amalgam
Transition metals with lowest melting point is:
In lake test for Al3+ ions, there is the formation of coloured ‘floating lake’. It is due to ______.
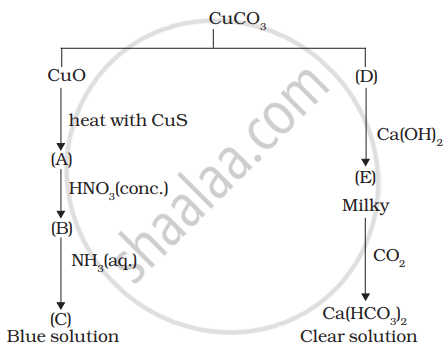
Identify A to E and also explain the reactions involved.
Identify the metal and justify your answer.
Carbonyl \[\ce{M(CO)5}\]
The element with atomic number 46 belongs to
Why Zn, Cd and Hg are not called transition metals?
Consider the following standard electrode potential values:
\[\ce{Sn^{2+}_{ (aq)} + 2e^- -> Sn_{(s)}}\]; E0 = −0.14 V
\[\ce{Fe^{3+}_{ (aq)} + e^- -> Fe^{2+}_{ (aq)}}\]; E0 = +0.77 V
What is the cell reaction and potential for the spontaneous reaction that occurs?
