Advertisements
Advertisements
प्रश्न
Hydrogen chloride gas is prepared in the laboratory by the action of concentrated sulphuric acid on sodium chloride.
State the method of collection of the gas formed above.
उत्तर
Upward displacement of water.
APPEARS IN
संबंधित प्रश्न
Identify the acid in the case:
The acid on mixing with lead nitrate solution produces a white precipitate, which is insoluble even on heating.
Give appropriate scientific reasons for The electrical conductivity of acetic acid is less in comparison to the electrical conductivity of dilute sulphuric acid at a given concentration.
Define the following term : Hygroscopic substance
Name the following:
The precipitate obtained by treating carbon with hot concentrated sulphuric acid.
What would you observe in the following case?
Dilute sulphuric acid is added to ferrous sulphate solution.
Give reason for the following:
Sulphuric acid can form two kinds of salts with sodium chloride.
Describe the reaction that show
Dilute sulphuric acid behaves as dibasic acid.
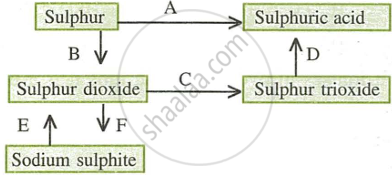
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
What would you observe in the following case:
Sugar crystals are added to a hard glass test tube containing concentrated sulphuric acid.
Give one equation to show the following properties of sulphuric acid:
As a non-volatile acid
