Advertisements
Advertisements
प्रश्न
Name the following:
The precipitate obtained by treating carbon with hot concentrated sulphuric acid.
उत्तर
When carbon is treated with hot conc. Sulphuric acid then water, sulphur dioxide and carbon dioxide are formed.
\[\ce{C+ 2H2SO4 ->2H2O + 2SO2 + CO2 }\]
APPEARS IN
संबंधित प्रश्न
Write balanced chemical equations for the action of dilute sulphuric acid on sodium sulphite.
The acid which is used in the preparation of a volatile acid
Give balanced chemical equations for the action of sulphuric acid on the following:
Sulphur
Name the following:
The precipitate obtained by treating aqueous barium chloride with dilute sulphuric acid.
Why is sulphuric acid known as king of chemicals and oil vitriol ?
Give examples of the use of sulphuric acid as
An oxidizing agent
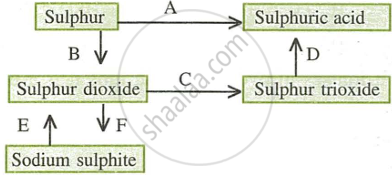
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Give balanced chemical equation to prepare the following salt:
Lead sulphate from lead carbonate
Identify the salts P and Q from the observation given below:
On performing the flame test salt P produces a lilac coloured flame and its solution gives a white precipitate with silver nitrate solution. Which is soluble in ammonium hydroxide solution.
An acid obtained from concentrated nitric acid on reaction with Sulphur ______.
