Advertisements
Advertisements
प्रश्न
Hydrogen gas is diatomic whereas inert gases are monoatomic – Explain on the basis of MO theory.
उत्तर
The molecular orbital electronic configuration of the hydrogen molecule is (σ1s2). The molecular orbital energy level diagram of the H2 molecule is given in
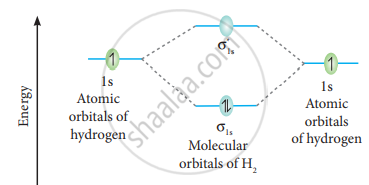
Here, N2 = 2, Na = 0
Bond order = `("N"_"b" - "N"_"a")/2`
`= (2 - 0)/2` = 1
He2: σ1s2 σ1s*2
The molecular orbital energy level diagram of He2 (hypothetical) is given in
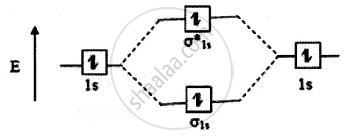
Here, Nb = 2 and Na = 2
Bond order = `("N"_"b" - "N"_"a")/2`
`(2 - 2)/2` = 0
As the bond order for He2 comes out to form between two hydrogen atoms. But as the bond order of helium is zero, there is no bond between helium atoms and hence it is monoatomic.
Result:
As the bond order of the H2 molecule is one, it is diatomic and a single bond is formed between two hydrogen atoms. But as the bond order of helium is zero, there is no bond between helium atoms and hence it is monoatomic.
APPEARS IN
संबंधित प्रश्न
Select and write the most appropriate alternatives from the given choices.
Which molecule is linear?
Select and write the most appropriate alternatives from the given choices.
Which O2 molecule is paramagnetic. It is explained on the basis of ______.
Draw an orbital diagram of Hydrogen fluoride molecule
BF3 molecule is planar but NH3 pyramidal. Explain.
CaO and NaCl have the same crystal structure and approximately the same radii. If U is the lattice energy of NaCl, the approximate lattice energy of CaO is ______.
Draw MO diagram of CO and calculate its bond order.
What do you understand by Linear combination of atomic orbitals in MO theory?
4p, 4d, 5s and 5p orbitals are arranged in the order of decreasing energy. The CORRECT option is:
Which one of the following sets CORRECTLY represents the increase in the paramagnetic property of the ions?
What is the bond order of B2 molecule?
