Advertisements
Advertisements
प्रश्न
Hydrogen gas is diatomic whereas inert gases are monoatomic – Explain on the basis of MO theory.
उत्तर
The molecular orbital electronic configuration of the hydrogen molecule is (σ1s2). The molecular orbital energy level diagram of the H2 molecule is given in
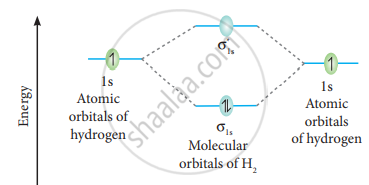
Here, N2 = 2, Na = 0
Bond order =
He2: σ1s2 σ1s*2
The molecular orbital energy level diagram of He2 (hypothetical) is given in
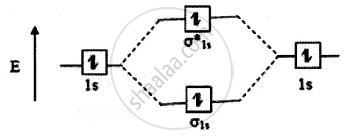
Here, Nb = 2 and Na = 2
Bond order =
As the bond order for He2 comes out to form between two hydrogen atoms. But as the bond order of helium is zero, there is no bond between helium atoms and hence it is monoatomic.
Result:
As the bond order of the H2 molecule is one, it is diatomic and a single bond is formed between two hydrogen atoms. But as the bond order of helium is zero, there is no bond between helium atoms and hence it is monoatomic.
APPEARS IN
संबंधित प्रश्न
Why is O2 molecule paramagnetic?
Pick out the incorrect statement from the following:
Draw MO diagram of CO and calculate its bond order.
Discuss the formation of N2 molecule using MO Theory.
Which of the following statements is INCORRECT?
Which one of the following sets CORRECTLY represents the increase in the paramagnetic property of the ions?
Among the following MOs, the highest energy MO that is occupied by electrons in case of N2 molecule is ____________.
Of the ions Ti4+, Co2+ and Cr3+, ____________.
Which element among the following does NOT form diatomic molecules?
Bonding in which of the following diatomic molecule(s) become(s) stronger, on the basis of MO Theory, by removal of an electron?
(A) NO
(B) N2
(C) O2
(D) C2
(E) B2
Choose the most appropriate answer from the options given below:
