Advertisements
Advertisements
प्रश्न
Identify a more favourable resonance structure from the following. Justify.
\[\begin{array}{cc}
\phantom{.............}\ce{O-}\phantom{......................}\ce{O+}\\\phantom{............}|\phantom{.........................}|\\
\ce{^{+}CH3 - CH = C - H ↔ ^{-}CH2 - CH = C - H}
\end{array}\]
उत्तर
\[\begin{array}{cc}
\phantom{.............}\ce{O-}\phantom{......................}\ce{O+}\\\phantom{............}|\phantom{.........................}|\\
\ce{^{+}CH3 - CH = C - H ↔ ^{-}CH2 - CH = C - H}
\end{array}\]
(I) (II)
Both structures (I) and (II) involves the separation of opposite charges, but structure (I) has a positive charge on the more electropositive ‘C’ and a negative charge on more electronegative ‘O’. Thus, structure (I) will be a more favourable resonance structure.
APPEARS IN
संबंधित प्रश्न
Match the pairs.
| Column 'A' | Column 'B' | ||
| i. | Inductive effect | a. | delocalisation of π electrons |
| ii. | Hyperconjugation | b. | displacement of π electrons |
| iii. | Resonance effect | c. | delocalisation of σ electrons |
| d. | displacement of σ electrons | ||
Write IUPAC names of the following.

Write the IUPAC name of the following.
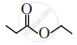
Observe the following structures and answer the questions given below.
- \[\ce{CH3 - CH2 - CH2 - CHO}\]
\[\begin{array}{cc}\ce{CH3 - CH - CHO}\\
|\phantom{...}\\\ce{CH3}\end{array}\]
a. What is the relation between (i) and (ii)?
b. Write IUPAC name of (ii).
c. Draw the functional group isomer of (i).
Phytane is a naturally occurring alkane produced by the alga spirogyra and is a constituent of petroleum. The IUPAC name for phytane is 2,6,10,14-tetramethylhexadecane. Write a zig-zag formula for phytane. How many primary, secondary, tertiary, and quaternary carbons are present in this molecule?
IUPAC name of \[\begin{array}{cc}
\phantom{....}\ce{H}\phantom{...}\ce{C4H9}\\
|\phantom{....}|\\\ce{CH3 - C - C - CH3}\\
|\phantom{....}|\\\phantom{.....}\ce{C2H5}\phantom{.}\ce{CH3}\phantom{...}\end{array}\] is
The IUPAC name of the compound\[\begin{array}{cc}\ce{CH3-CH=C-CH2-CH3}\\
|\phantom{..}\\\phantom{...............}\ce{CH2 - CH2 - CH3}\end{array}\] is
Give the IUPAC names of the following compound.
\[\ce{CH3 - O - CH3}\]
Give the IUPAC names of the following compound.
\[\begin{array}{cc}\ce{CH3 - CH2 - CH - CHO}\\
\phantom{.....}|\\\phantom{.......}\ce{OH}
\end{array}\]
Give the IUPAC names of the following compound.
\[\ce{CH2 = CH - CH = CH2}\]
Give the IUPAC names of the following compound.
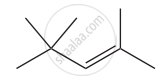
Give the structure for the following compound.
3 – ethyl – 2 methyl – 1 – pentene
Give the structure for the following compound.
tertiary butyl iodide
Give the structure for the following compound.
3 - Chlorobutanal
Give the structure for the following compound.
2 - Chloro - 2- methyl propane
Give the structure for the following compound.
2,2-dimethyl-1-chloropropane
Give the structure for the following compound.
3 - methylbutan - 2 - ol
As per IUPAC nomenclature, the name of the complex Na3[AlF6] is ____________.
An organic compound on treatment with concentrated NaOH gave 2, 2-dimethylpropan-1-ol and sodium salt of 2, 2-dimethylpropanoic acid. The compound is:
In the reaction, \[\ce{Anisole + {'A'} ->[Anhydrous][AlCl3] 4-Methoxyacetophenone}\]
'A' is ____________.
What is the IUPAC name of the following compound?
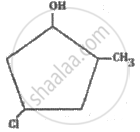
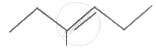
The IUPAC name of the following compound is:
What is a common name of the compound 1-Chloro-2, 2-dimethylpropane?
In the trivial system which prefix will be used for the following compound?
\[\begin{array}{cc}
\ce{CH3}\phantom{.}\\
\phantom{...}\backslash\\
\ce{CH3 - C -}\\
\phantom{...}/\\
\ce{CH3}\phantom{.}\end{array}\]
Consider the following reactions.
\[\ce{C6H5N^+_2Cl- ->[HBF4] A ->[NaNO2/Cu] B ->[Sn/HCl] C}\]
Identify the compound formed C in the above reaction.
Write the structural formulae for the following name and also write the correct IUPAC names for that.
2,2,3-trimethylpentan-4-ol
Write the structural formulae for the following name and also write the correct IUPAC name for that.
2,2,3-trimethylpentan-4-ol
