Advertisements
Advertisements
Question
Identify a more favourable resonance structure from the following. Justify.
\[\begin{array}{cc}
\phantom{.............}\ce{O-}\phantom{......................}\ce{O+}\\\phantom{............}|\phantom{.........................}|\\
\ce{^{+}CH3 - CH = C - H ↔ ^{-}CH2 - CH = C - H}
\end{array}\]
Solution
\[\begin{array}{cc}
\phantom{.............}\ce{O-}\phantom{......................}\ce{O+}\\\phantom{............}|\phantom{.........................}|\\
\ce{^{+}CH3 - CH = C - H ↔ ^{-}CH2 - CH = C - H}
\end{array}\]
(I) (II)
Both structures (I) and (II) involves the separation of opposite charges, but structure (I) has a positive charge on the more electropositive ‘C’ and a negative charge on more electronegative ‘O’. Thus, structure (I) will be a more favourable resonance structure.
APPEARS IN
RELATED QUESTIONS
Identify a more favourable resonance structure from the following. Justify.

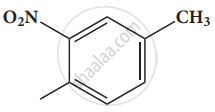
Write the IUPAC name of the following.
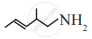
Write the IUPAC name of the following.
The IUPAC name of the compound\[\begin{array}{cc}\ce{CH3-CH=C-CH2-CH3}\\
|\phantom{..}\\\phantom{...............}\ce{CH2 - CH2 - CH3}\end{array}\] is
Give the IUPAC names of the following compound.
\[\ce{CH3 - O - CH3}\]
Give the IUPAC names of the following compound.
\[\begin{array}{cc}\ce{CH3 - CH2 - CH - CHO}\\
\phantom{.....}|\\\phantom{.......}\ce{OH}
\end{array}\]
Give the IUPAC names of the following compound.
\[\ce{CH2 = CH - CH = CH2}\]
Give the IUPAC names of the following compound.
\[\begin{array}{cc}\ce{CH3 - C ≡ C - CH - CH3}\\
\phantom{........}|\\\phantom{.........}\ce{Cl}
\end{array}\]
Give the IUPAC names of the following compound.
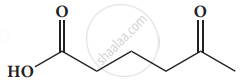
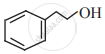
Give the IUPAC names of the following compound.
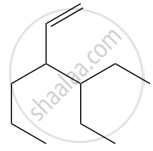
Give the IUPAC names of the following compound.
Give the structure for the following compound.
tertiary butyl iodide
Give the structure for the following compound.
3 - Chlorobutanol
Give the structure for the following compound.
2 - Chloro - 2- methyl propane
Give the structure for the following compound.
3 - methylbut -1- ene
Give the structure for the following compound.
acetaldehyde
Which among the following carbocation is most reactive?
As per IUPAC nomenclature, the name of the complex [Fe(H2O)5(NCS)]2+ is ____________.
lUP AC name of
\[\begin{array}{cc}
\phantom{}\ce{C2H5}\phantom{.}\ce{Cl}\phantom{....}\ce{CH3}\phantom{......}\\
\phantom{}|\phantom{.....}|\phantom{......}|\phantom{........}\\
\ce{H3C - CH2 - CH - CH - CH - CH2 - CH2 - CH3}
\end{array}\]
An organic compound on treatment with concentrated NaOH gave 2, 2-dimethylpropan-1-ol and sodium salt of 2, 2-dimethylpropanoic acid. The compound is:
Which reagent can be used to convert 1-nitropropane to propan-1-amine?
What is a common name of the compound 1-Chloro-2, 2-dimethylpropane?
The IUPAC name of the following compound is:
CH2 = C = CH − CH3
The IUPAC name for the following compound is:
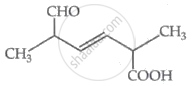
The IUPAC name for the following compound is:
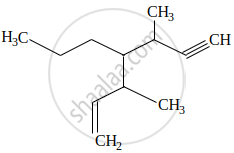
The IUPAC name of the following compound is:
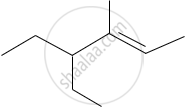
Write the structural formulae for the following name and also write the correct IUPAC name for them.
2,2,3-trimethylpentan-4-ol
