Advertisements
Advertisements
प्रश्न
Identify the process given in following passage and draw neat labelled diagram showing the process.
Electrolysis of molten mixture of alumina (melting point > 20000C) is done in a steel tank. The tank has a graphite lining on the inner side. This lining does the work of a cathode. A set of graphite rods dipped in the molten electrolyte works as anode. Cryolite (Na3AlF6) and fluorspar (CaF2) are added in the mixture to lower its melting point upto 10000C.
उत्तर
Electrolytic reduction of Alumina :
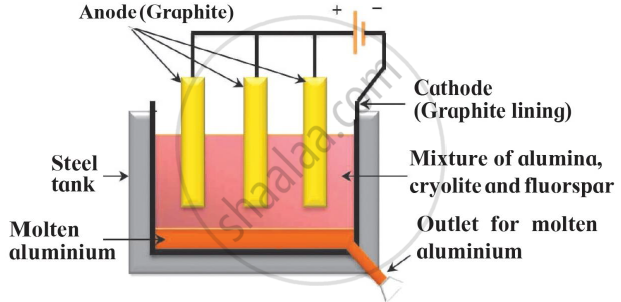
APPEARS IN
संबंधित प्रश्न
Name two metals which are found in nature in the free state.
In the extraction of aluminium: Draw the diagram for the extraction of aluminium.
Name one metal which is extracted by reduction with carbon.
Explain why, an aqueous solution of sodium chloride is not used for the electrolytic extraction of sodium metal.
Name one ore of aluminium. Name the aluminium compound present in this ore and write its chemical formula.
Calamine ore can be used to extract one of the following metals. This metal is:
(a) copper
(b) mercury
(c) aluminium
(d) zinc
The articles made of silver metal become dark on prolonged exposure to air. This is due to the formation of a layer of its:
(a) oxide
(b) hydride
(c) sulphide
(d) carbonate
State three objectives achieved during the roasting of ores.
Give the principles of the hydrolytic method.
Aluminum is used in thermite welding:
write reaction for process?
Name the anode, the cathode and the electrolyte used in the electrolytic refining of impure copper.
Define the following term.
Mineral
What is meant by concentration of ores?
Compare the process of calcination and roasting.
Choose the correct answer from the options given below:
The metal is a liquid at room temperature.
Complete the following by selecting the correct option from the choices given:
The metal which does not react with water or dilute H2SO4 but reacts with concentrated H2SO4 is _________
Usually ______ ores are subjected to ______ which is done in the absence of air.
Atomic number of aluminium is _______ and its electronic configuration is _______.
Write the molecular formulae of the following compound.
Sodium aluminate
Which of the following metals exist in their native state in nature?
(i) Cu
(ii) Au
(iii) Zn
(iv) Ag
