Advertisements
Advertisements
प्रश्न
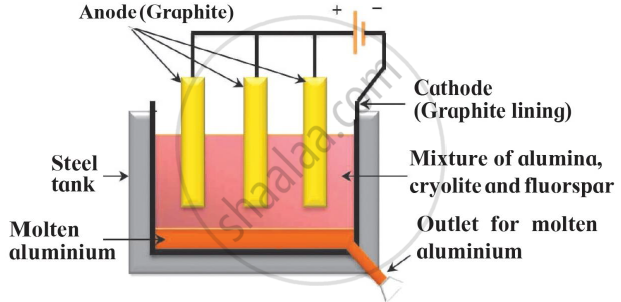
Identify the process given in following passage and draw neat labelled diagram showing the process.
Electrolysis of molten mixture of alumina (melting point > 20000C) is done in a steel tank. The tank has a graphite lining on the inner side. This lining does the work of a cathode. A set of graphite rods dipped in the molten electrolyte works as anode. Cryolite (Na3AlF6) and fluorspar (CaF2) are added in the mixture to lower its melting point upto 10000C.
उत्तर
Electrolytic reduction of Alumina :
APPEARS IN
संबंधित प्रश्न
Name the gas in air which tarnishes silver articles slowly.
Explain giving one example, how highly reactive metals (which are high up in the reactivity series) are extracted.
Define the term mineral.
Name the electrode at which aluminium metal is produced.
Which metal is extracted from calamine ore?
In an electrolytic tank, aluminium metal is being extracted by the electrolysis of molten aluminium oxide using carbon electrodes. It is observed that one of the carbon electrodes is gradually burnt away and has to be replaced.
(a) Which carbon electrode (cathode or anode) is burnt away?
(b) Why is this carbon electrode burnt away?
Some metallic oxides can be reduced by hydrogen, carbon and carbon monoxide and some cannot. Explain.
Name the ore of mercury. With the help of balanced chemical equations, explain the process of extraction of mercury from its ore.
What is passive iron?
Arrange the metals of copper, iron, magnesium, sodium and zinc in the decreasing order of reactivity.
How is the method of extraction of metals high up in the reactivity series different from that for metals in the middle? Why can the same process not be applied for them? Name the process used for the extraction of these metals.
Iron is _______.
Write the name.
Metals which are amphoteric in nature.
Mercury, silver, gold are highly reactive metals.
Explain the following reaction with the balanced equation.
Ferric oxide is reacted with aluminium.
Complete flow chart given below.
In thermite welding a mixture of ____________ is ignited with a burning magnesium ribbon which produces molten iron metal as a large amount of heat is evolved.
Electrical wires have a coating of an insulting material. The material, generally used is
