Advertisements
Advertisements
प्रश्न
Explain giving one example, how highly reactive metals (which are high up in the reactivity series) are extracted.
उत्तर
Sodium, potassium, calcium, magnesium and aluminium are highly reactive metals and are placed at the top of the reactivity series. These metals are extracted by electrolytic reduction of their molten chlorides or oxides because these metals are not reduced by other reducing agents like coke, carbon monoxide etc.
Let us take an example of extraction of sodium metal. Sodium metal is extracted by electrolytic reduction of molten sodium chloride. On passing electricity through molten sodium chloride, decomposition reaction occurs and formation of sodium metal and chlorine gas takes place.
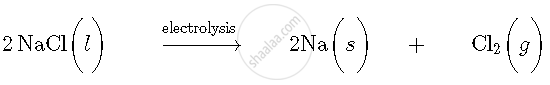
Molten sodium chloride consists of sodium and chloride ions. The reactions that occurs during electrolysis are:
1. Cathode produces electrons to reduce sodium ions to sodium atoms (or sodium metal) by acting as a reducing agent. Sodium ions are cations; therefore, they get attracted to negatively charged cathode and get deposited there.
Cathode: 2Na+ + 2e− → 2Na
Sodium ions Electrons Sodium atoms
(from molten NaCl) from cathode (or sodium metal)
2. Chloride ions, being anions, get attracted to positively charged anode. These chloride ions are oxidised to chlorine gas. Chlorine gas is produced at anode.
Anode: 2Cl− − 2e− → Cl2
Chloride ions electrons Chlorine gas
(from molten NaCl) to anode
APPEARS IN
संबंधित प्रश्न
Name one metal which is extracted by reduction with heat alone.
Name two metals which are always found in combined state.
What is the difference between a mineral and an ore?
Give the name of one ore of iron. Which iron compound is present in this ore? Write its chemical formula.
Give reasons, why aluminum is used in:
making alloys
Identify the process given in following passage and draw neat labelled diagram showing the process.
Electrolysis of molten mixture of alumina (melting point > 20000C) is done in a steel tank. The tank has a graphite lining on the inner side. This lining does the work of a cathode. A set of graphite rods dipped in the molten electrolyte works as anode. Cryolite (Na3AlF6) and fluorspar (CaF2) are added in the mixture to lower its melting point upto 10000C.
Name the following:
The process in which an ore is heated in air so that oxygen gets added to it to form the oxides.
To protect iron from rusting, it is coated with a thin layer of zinc. Name the process.
During extraction of metals, electolytic refining is used to obtain pure metals.
(a) Which material will be used as anode and cathode for refining of silver metal by this process?
(b) Suggest a suitable electrolyte also.
(c) In this electrolytic cell, where do we get pure silver after passing electric current?
A mineral from which the metal can be extracted economically and conveniently is known as ______.
