Advertisements
Advertisements
प्रश्न
Describe with one example, how moderately reactive metals (which are in the middle of reactivity series) are extracted.
उत्तर
The oxides of moderately reactive metals like zinc, iron, lead copper, placed in the middle of the reactivity series, are reduced by carbon because carbon is more reactive.
Let us take an example of extraction of zinc from its carbonate ore, i.e., calamine.
The steps involved in the extraction of concentrated calamine ore are:
1. Carbonate of zinc is converted to its oxide by calcination process. In calcination process, zinc carbonate ore is heated strongly in the absence of air to produce zinc oxide and carbon dioxide gas

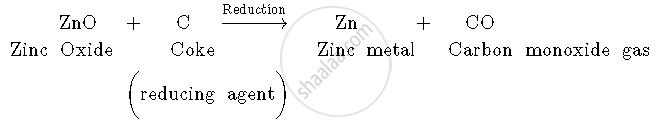
2. Zinc oxide is then reduced by coke (carbon). When zinc oxide is heated with coke, carbon acts as a reducing agent and reduces zinc oxide to zinc metal.
APPEARS IN
संबंधित प्रश्न
Why does aluminium not corrode right through?
Explain why, an aqueous solution of sodium chloride is not used for the electrolytic extraction of sodium metal.
Give the principles of the hydrolytic method.
Some metallic oxides can be reduced by hydrogen, carbon and carbon monoxide and some cannot. Explain.
Aluminum is used in thermite welding:
what is ignition mixture?
Aluminum is used in thermite welding:
write reaction for process?
Name the following:
Name two metals always find in combined state.
Name : The metal which is liquid at room temperature.
Write the molecular formulae of the following compound.
Sodium aluminate
Explain the following reaction with the balanced equation.
Dry aluminium hydroxide is ignited at 1000 °C
