Advertisements
Advertisements
प्रश्न
Describe with one example, how moderately reactive metals (which are in the middle of reactivity series) are extracted.
उत्तर
The oxides of moderately reactive metals like zinc, iron, lead copper, placed in the middle of the reactivity series, are reduced by carbon because carbon is more reactive.
Let us take an example of extraction of zinc from its carbonate ore, i.e., calamine.
The steps involved in the extraction of concentrated calamine ore are:
1. Carbonate of zinc is converted to its oxide by calcination process. In calcination process, zinc carbonate ore is heated strongly in the absence of air to produce zinc oxide and carbon dioxide gas

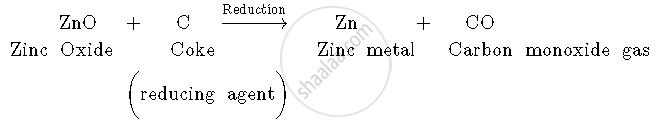
2. Zinc oxide is then reduced by coke (carbon). When zinc oxide is heated with coke, carbon acts as a reducing agent and reduces zinc oxide to zinc metal.
APPEARS IN
संबंधित प्रश्न
In the extraction of aluminium: Name the process of concentration of bauxite.
In the extraction of aluminium: Draw the diagram for the extraction of aluminium.
Define the following term.
Gangue
State one observation for each of the following :
Copper sulphate solution is electrolysed using copper electrodes.
Name the following:
The substance added to get rid of gangue in the extraction of metal.
Observe the given figure of reactivity series of metals and answer the following questions:

Reactivity series of metals
- Name two metals which react with water.
- Name two moderately reactive metals.
- Name the most highly reactive metal and the most less reactive metal.
Extraction of moderately reactive elements is done by _______ and _______ method.
Write the name.
Metals which are amphoteric in nature.
Write the name.
The process of extraction of aluminium from alumina-
Care must be taken while diluting concentrated nitric acid with water. Why?
