Advertisements
Advertisements
प्रश्न
If the electronegativity difference between two bonded atoms in a molecule is greater than 1.7, the nature of bonding is ______
उत्तर
If the electronegativity difference between two bonded atoms in a molecule is greater than 1.7, the nature of bonding is an Ionic bond.
APPEARS IN
संबंधित प्रश्न
Name the elements with highest and lowest electronegativity ?
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 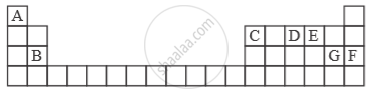
Which element forms electrovalent compound with G?
Among the elements of the second period, Li to Ne, pick out the element with the highest electro negativity
Fill in the blank
The maximum electro negativity is shown by ______.
Select the correct answer
Which of the following element has highest electronegativity?
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (decreasing electronegativity)
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The electronegativity:
Identify the following:
The most electronegative element of Period 3.
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (decreasing electronegativity)
In the table below, H does not represent hydrogen. Some elements are given in their own symbol and position in the periodic table while others are shown with a letter.
| IA | IIA | IIIA | IVA | VA | VIA | VIIA | O |
| 1 | 2 | 13 | 14 | 15 | 16 | 17 | 18 |
| Li | D | O | J | Ne | |||
| A | Mg | E | Si | H | K | ||
| A | C | F | G | L |
Select from the table.
- Which is most electronegative.
- How many valence electrons are present in G.
- Write the formula of the compound between B and H.
- In the compound between F and J what type of bond will be formed.
- Draw the electron dot structure for the compound formed between C and K.
