Advertisements
Advertisements
प्रश्न
In the laboratory preparation of hydrochloric acid, \[\ce{HCl}\] gas is dissolved in water.
- Draw a diagram to show the arrangement used for the absorption of \[\ce{HCl}\] gas in water.
- State why such an arrangement is necessary? Give two reasons for the same.
- Write the chemical equation for the laboratory preparation of \[\ce{HCl}\] gas when the reactants are:
- Below 200°C
- Above 200°C
उत्तर
i. 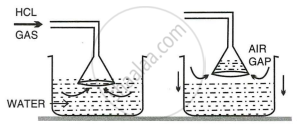
ii. Two reasons for the use of funnel arrangements are:
- Prevents or minimizes back-suction of water.
- Provides a large surface area for absorption of \[\ce{HCl}\] gas.
iii.
- \[\ce{NaCl + H2SO4 ->[below 200^{\circ}C] NaHSO4 + HCl ^}\]
- \[\ce{2NaCl + H2SO4 ->[above 200^{\circ}C] Na2SO4 + 2HCl}\]
APPEARS IN
संबंधित प्रश्न
Explain, why (or give reasons for)
Hydrochloric acid cannot be concentrated above 22.2% by boiling.
Write three equations to show that hydrochloric acid acts as an acid.
Describe an experiment to prove the following:
HCI is highly soluble in water.
Name the gas evolved when an oxide and concentrated hydrochloric acid are heated.
Write equation for the reaction between hydrochloric acid of the following: Bleaching powder
Write a balanced equation for the reaction of zinc and dilute hydrochloric acid.
Aqua regia is a mixture of ______.
State one appropriate observation of the following:
Which gas is evolved when potassium sulphite with dilute hydrochloric acid.
State your observation when dilute hydrochloric acid is added to sodium thisulphate.
Name the anion present in the following compound:
The action of heat on the insoluble compound C produces a gas which turns lime water turbid.
