Advertisements
Advertisements
Question
In the laboratory preparation of hydrochloric acid, \[\ce{HCl}\] gas is dissolved in water.
- Draw a diagram to show the arrangement used for the absorption of \[\ce{HCl}\] gas in water.
- State why such an arrangement is necessary? Give two reasons for the same.
- Write the chemical equation for the laboratory preparation of \[\ce{HCl}\] gas when the reactants are:
- Below 200°C
- Above 200°C
Solution
i. 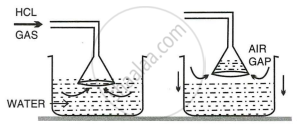
ii. Two reasons for the use of funnel arrangements are:
- Prevents or minimizes back-suction of water.
- Provides a large surface area for absorption of \[\ce{HCl}\] gas.
iii.
- \[\ce{NaCl + H2SO4 ->[below 200^{\circ}C] NaHSO4 + HCl ^}\]
- \[\ce{2NaCl + H2SO4 ->[above 200^{\circ}C] Na2SO4 + 2HCl}\]
APPEARS IN
RELATED QUESTIONS
How will you prepare hydrochloric acid in laboratory? OR
Describe briefly the method employed to dissolve hydrogen chloride gas in water as it is prepared. What is the purpose of funnel while preparing hydrochloric acid from HCI gas?
What would happen if
Hydrogen chloride prepared in laboratory, is passed through water, using a delivery tube.
Write three equations to show that hydrochloric acid acts as an acid.
Fill in the blank:
Hydrogen and chlorine reacts in presence of ___________ to form hydrogen chloride.
Write equation for the reaction between hydrochloric acid of the following: Manganese dioxide
Write a balanced equation for the reaction of zinc and dilute hydrochloric acid.
Correct the following statement:
Hydrochloric acid is prepared in the laboratory by passing hydrogen chloride directly through water.
State one appropriate observation of the following:
Action of dilute HCl or sodium sulphite.
The following question is pertaining to the laboratory pertaining hydrogen chloride gas.
State a safety precaution you would take during the preparation of hydrochloric acid.
State your observation when dilute hydrochloric acid is added to sodium thisulphate.
