Advertisements
Advertisements
प्रश्न
In the laboratory preparation of hydrochloric acid, \[\ce{HCl}\] gas is dissolved in water.
- Draw a diagram to show the arrangement used for the absorption of \[\ce{HCl}\] gas in water.
- State why such an arrangement is necessary? Give two reasons for the same.
- Write the chemical equation for the laboratory preparation of \[\ce{HCl}\] gas when the reactants are:
- Below 200°C
- Above 200°C
उत्तर
i. 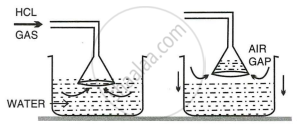
ii. Two reasons for the use of funnel arrangements are:
- Prevents or minimizes back-suction of water.
- Provides a large surface area for absorption of \[\ce{HCl}\] gas.
iii.
- \[\ce{NaCl + H2SO4 ->[below 200^{\circ}C] NaHSO4 + HCl ^}\]
- \[\ce{2NaCl + H2SO4 ->[above 200^{\circ}C] Na2SO4 + 2HCl}\]
APPEARS IN
संबंधित प्रश्न
Explain, why (or give reasons for)
A solution of HCl gas in water turns blue litmus red and conducts electricity, while HCI gas dissolves in toluene and it has no effect on litmus and does not conduct electricity.
What would happen if :
Concentrated hydrochloric acid is kept open.
What would happen if
Hydrogen chloride prepared in laboratory, is passed through water, using a delivery tube.
Give two tests for hydrochloric acid.
Fill in the blank:
___________ agent converts hydrochloric acid to chlorine.
Fill in the blank:
The white precipitate of lead chloride is soluble in _________
Why is hydrogen chloride not collected over water?
Aqua regia is a mixture of ______.
State one appropriate observation of the following :
Copper sulphate is treated with dilute hydrochloric acid
State your observation when dilute hydrochloric acid is added to sodium thisulphate.
