Advertisements
Advertisements
प्रश्न
In the cubic close packing, the unit cell has ______.
विकल्प
4 tetrahedral voids each of which is shared by four adjacent unit cells.
4 tetrahedral voids within the unit cell.
8 tetrahedral voids each of the which is shared by four adjacent unit cells.
8 tetrahedral voids within the unit cells.
उत्तर
In the cubic close packing, the unit cell has 8 tetrahedral voids within the unit cells.
Explanation:
Eight tetrahedral voids per fee unit cell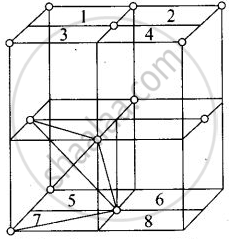
Each cube represented by numeric 1, 2, 3, 4, 5, 6, 7, 8 contains one tetrahedral void.
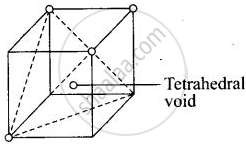
Each cube contains one tetrahedral void at its body centre as shown above
APPEARS IN
संबंधित प्रश्न
If the radius of the octachedral void is r and radius of the atoms in close packing is R, derive relation between r and R.
The unit cell of a substance has cations A+ at the corners of the unit cell and the anions B− at the center. The simplest formula of the substance is ____________.
What is the coordination number of sodium in Na2O?
The Ca2+ and F– are located in CaF2 crystal, respectively at face centered cubic lattice points and in ____________.
If AgI crystallises in zinc blende structure with I– ions at lattice points. What fraction of tetrahedral voids is occupied by Ag+ ions?
The number of octahedral voids present in a lattice is A. The number of closed packed articles, the number of tetrahedral voids generated is B the number of closed packed particles:
If Germanium crystallises in the same way as diamond, then which of the following statement is not correct?
In which of the following arrangements octahedral voids are formed?
(i) hcp
(ii) bcc
(iii) simple cubic
(iv) fcc
The coordination number of Y will be in the XY types of crystal:
A solid compound XY has Nacl structure. If the radium of cation (X+) is 100 pm, the radium of anion (r–) will be:-
