Advertisements
Advertisements
Question
In the cubic close packing, the unit cell has ______.
Options
4 tetrahedral voids each of which is shared by four adjacent unit cells.
4 tetrahedral voids within the unit cell.
8 tetrahedral voids each of the which is shared by four adjacent unit cells.
8 tetrahedral voids within the unit cells.
Solution
In the cubic close packing, the unit cell has 8 tetrahedral voids within the unit cells.
Explanation:
Eight tetrahedral voids per fee unit cell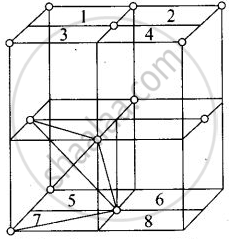
Each cube represented by numeric 1, 2, 3, 4, 5, 6, 7, 8 contains one tetrahedral void.
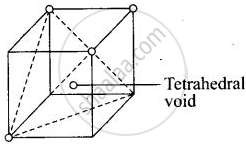
Each cube contains one tetrahedral void at its body centre as shown above
APPEARS IN
RELATED QUESTIONS
What is the formula of a compound in which the element Y forms hcp lattice and atoms of X occupy 2/3rd of tetrahedral voids?
The unit cell of a substance has cations A+ at the corners of the unit cell and the anions B− at the center. The simplest formula of the substance is ____________.
Total number of voids in 0.5 mole of a compound which forms hexagonal close packed structure is ____________.
A compound is formed by two elements A and B. The element B forms cubic close packing and atoms of A occupy `1/3"rd"` of tetrahedral voids. The formula of this compound is ____________.
What is the coordination number of sodium in Na2O?
If AgI crystallises in zinc blende structure with I– ions at lattice points. What fraction of tetrahedral voids is occupied by Ag+ ions?
Which set of following characteristics for ZnS crystal is correct?
The number of tetrahedral voids per unit cell in NaCl crystal is:
(i) 4
(ii) 8
(iii) twice the number of octahedral voids.
(iv) four times the number of octahedral voids.
In a compound, nitrogen atoms (N) make cubic close packed lattice and metal atoms (M) occupy one-third of the tetrahedral voids present. Determine the formula of the compound formed by M and N?
In which of the following structures coordination number for cations and anions in the packed structure will be same?
