Advertisements
Advertisements
प्रश्न
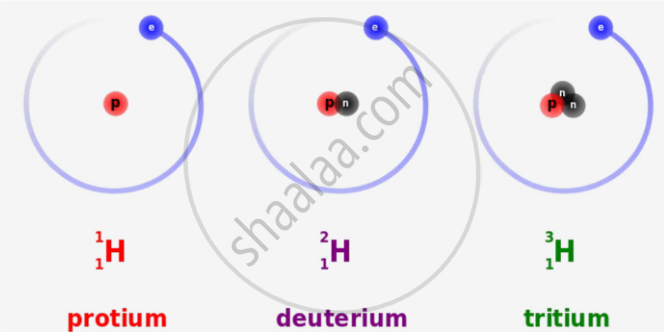
In what respects do the three isotopes of hydrogen differ? Give their structures.
उत्तर
The three isotopes differ only due to their mass number which is respectively 1, 2 and 3 and named protium, deuterium and tritium.
APPEARS IN
संबंधित प्रश्न
The average atomic mass of a sample of an element X is 16.2 u. What are the percentages of isotopes `""_8^16X" and """_8^18X` in the sample?
ANSWER IN DETAIL
State the postulates of the Dalton’s atomic theory.
What does an atom consist of ? Explain.
Define: Covalent bond
An atom has 13 protons and 14 neutrons.
What is the atomic number of the atom ?
Define: An Atom:
Write the full form of IUPAC.
An atom of an element X may be written as `""_9^4"X"`
(a) What does the figure 9 indicate?
(b) What does the figure 4 indicate?
(c) What is the number of protons in atom X?
(d) What is the number of neutrons in atom X?
(e) What is the number of electrons in atom X?
(f) How many electrons are there in the outermost shell of an atom of element X?
(g) Write the symbol of ion formed by an atom of element X.
A sample of vitamic C is known to contain 2.58 ×1024 oxygen atoms. How many moles of oxygen atoms are present in the sample?
The graphite refill used is pencil is made of element called ______.
