Advertisements
Advertisements
प्रश्न
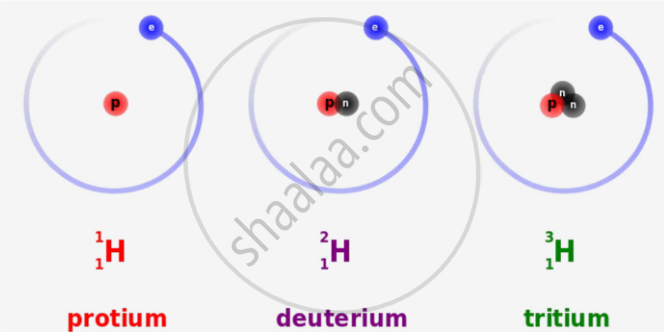
In what respects do the three isotopes of hydrogen differ? Give their structures.
उत्तर
The three isotopes differ only due to their mass number which is respectively 1, 2 and 3 and named protium, deuterium and tritium.
APPEARS IN
संबंधित प्रश्न
An atom has 13 protons and 14 neutrons.
What is the atomic number of the atom ?
An atom has 13 protons and 14 neutrons.
What is the atomic mass of the atom ?
A certain particle ‘X’ has 17 protons, 18 electrons and 20 neutrons.
What is the atomic numebr of X ?
Fill in the blank
An _______ is electrically neutral.
Draw diagrams representing the atomic structures of the following:
- Sodium atom
- Chlorine ion
- Carbon atom
- Oxygen ion
Define the following term with an example.
Metal
Which of the following statements made by Dalton has not undergone any change?
Atoms of one element can be transmuted into atoms of other elements by ______
1 mole of Gold and Silver contain same number of atoms.
Dalton’s atomic theory successfully explained
(i) Law of conservation of mass
(ii) Law of constant composition
(iii) Law of radioactivity
(iv) Law of multiple proportions
