Advertisements
Advertisements
Question
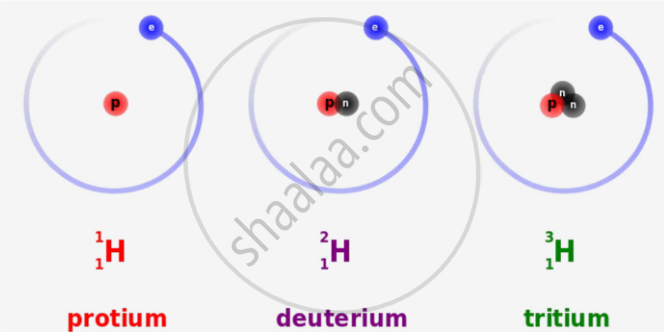
In what respects do the three isotopes of hydrogen differ? Give their structures.
Solution
The three isotopes differ only due to their mass number which is respectively 1, 2 and 3 and named protium, deuterium and tritium.
APPEARS IN
RELATED QUESTIONS
If bromine atom is available in the form of, say, two isotopes `""_35^79Br (49.7%)" and """_35^81Br (50.3%)`, calculate the average atomic mass of bromine atom.
WRITE SHORT ANSWER
What is radioactivity?
Fill in the blank:
Atoms are _____
What does the word ‘atom’ mean?
Name the three subatomic particles present in an atom.
Which of the following statement is correct about the atom of an element ?
Elements X and Y have 1 and 7 electrons in their outermost shell respectively.
Which will form negative ion?
Select the correct answer to the statement given below:
The basic unit of matter is not capable of independent existence.
Define the following term with an example.
Compound
The volume occupied by 1 mole of a diatomic gas at S.T.P is ______
