Advertisements
Advertisements
प्रश्न
Name any two electromagnetic waves which have a frequency higher than that of violet light. State one use of each.
उत्तर
The two electromagnetic waves and their uses having a frequency higher than that of violet light is:
X-rays: X-rays are very extensively used for medical purposes, e.g., in surgery to detect bone fractures by studying X-rays photographs.
Gamma rays: Gamma radiations are used in the cure of cancer.
APPEARS IN
संबंधित प्रश्न
To which part of the electromagnetic spectrum does a wave of frequency 5 × 1011 Hz belong?
Give a reason for the following:
It is necessary to use satellites for long-distance TV transmission. Why?
The small ozone layer on top of the stratosphere is crucial for human survival. Why?
Name the region beyond the red end of the spectrum.
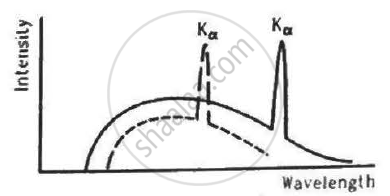
The figure shows the intensity-wavelength relations of X-rays coming from two different Coolidge tubes. The solid curve represents the relation for the tube A in which the potential difference between the target and the filament is VA and the atomic number of the target material is ZA. These quantities are VB and ZB for the other tube. Then,
50% of the X-rays coming from a Coolidge tube are able to pass through a 0.1 mm thick aluminium foil. If the potential difference between the target and the filament is increased, the fraction of the X-rays passing through the same foil will be
The Kβ X-ray of argon has a wavelength of 0.36 nm. The minimum energy needed to ionize an argon atom is 16 eV. Find the energy needed to knock out an electron from the K shell of an argon atom.
Find the maximum potential difference which may be applied across an X-ray tube with tungsten target without emitting any characteristic K or L X-ray. The energy levels of the tungsten atom with an electron knocked out are as follows.
| Cell containing vacancy | K | L | M |
| Energy in keV | 69.5 | 11.3 | 2.3 |
Name two sources of ultraviolet radiation.
Choose the correct option.
How does the frequency of a beam of ultraviolet light change when it travels from air into glass?
